The Nobel Prize for Chemistry in 2020 has been given to two women scientists Emmanuelle Charpentier and Jennifer Doudna for developing the CRISPR/Cas9 method that can change the DNA. It is a revolutionary technology that can contribute to new cancer therapies and the ability to cure inherited diseases.
A young girl with leukemia receiving chemotherapy.
Photo by National Cancer Institute on Unsplash
The story started three billion years ago. We think of bacteria as sources of infection, but bacteria themselves are also susceptible to being infected by viruses. So about three billion years ago, bacteria evolved a defense mechanism to fight viral infection. That defense mechanism is now better known as CRISPR.
The warhead in CRISPR is this blue protein (shown in the pic below) that acts like molecular scissors to cut the DNA, breaking the double helix structure into two pieces. If CRISPR could not distinguish between bacterial and viral DNA, it would not be a very useful defense system.
The most fascinating characteristic of CRISPR is that it can be programmed to search, bind, and cut only a selected DNA sequence. So when a bacterium encounters a virus for the first time, it can store a small snippet of the virus’s DNA for use as a program to direct the CRISPR scissors to chop that viral DNA sequence during a future infection. Cutting the virus’s DNA messes up the function of the cut viral gene and therefore disrupts the virus’s life cycle.
Remarkable researchers including Emmanuelle Charpentier, George Church, Jennifer Doudna, and Feng Zhang showed seven years ago how CRISPR scissors could be programmed to cut DNA sequences of our choosing instead of the viral DNA sequences chosen by bacteria.
Mechanism
CRISPR/Cas9 system consists of two components: the Cas9 and a guide RNA. Cas9 protein that can cut DNA and a guide RNA that can recognize the sequence of the DNA to be edited.
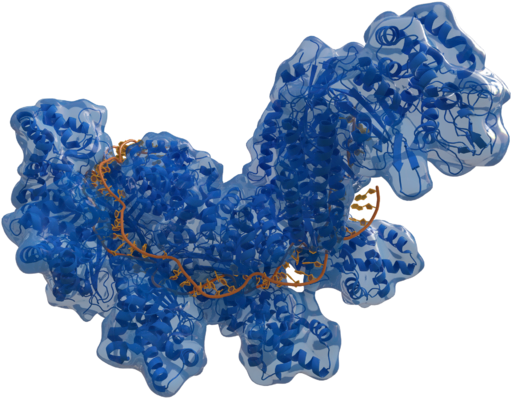
Pic Source: Creative Commons
Pic Source: Creative Commons
Scientists first identify the sequence of the human genome that is causing a health problem. Then they create a specific guide RNA to recognize that particular sequence. The guide RNA gets attached to the DNA cutting Cas9. It is then introduced to the target cells. In the cells, it locates the target sequence and cuts the DNA. After cutting the DNA, scientists can edit the existing genome by either modifying, deleting, or inserting a new sequence. It makes a CRISPR/Cas 9 cut-paste tool for DNA editing.
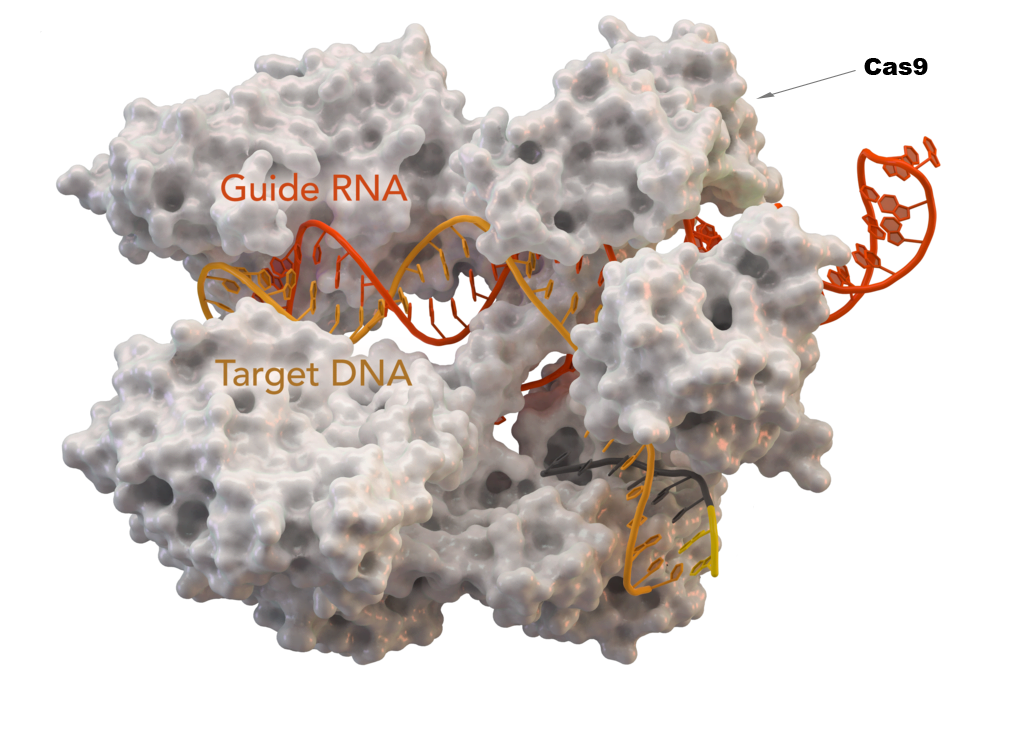
Pic Source: Creative Commons
CRISPR to the next level
DNA, the basic molecule forming the building blocks of every living organism, is composed of four bases: Adenine (A), Thymine (T), Cytosine (C), and Guanine (G). They are linked together in various combinations of base pairs and determine the characteristics of the genes.
Unhealthy eating, smoking, sunlight, even spontaneous mistakes made by our cells, cause changes in our genome. The common kind of change in DNA is the simple change of one letter such as C, with A, G, or T. These are called mutations.
Most of these mutations are harmless. But now and then, a point mutation causes a cell to misbehave in harmful ways or disrupts a crucial capability in a cell. If that mutation is inherited from your parents or occurs early enough in your development, then the result would be that many or all of your cells contain this harmful mutation. Then you would be one of the hundreds of millions of people with a genetic disease such as sickle cell anemia or progeria or muscular dystrophy etc.
We often know the point mutation or precise single letter change that causes the genetic disease and, in theory, could cure the disease.
But for most point mutations that cause genetic diseases simply cutting the already mutated gene will not benefit patients because the function of the mutated gene needs to be restored, not further disrupted. And while we can sometimes introduce new DNA sequences into cells to replace the DNA sequences surrounding a cut site, that process, unfortunately, does not work in most types of cells, and disrupted gene outcomes still predominate.

It quickly became clear that the popular gene-editing tool wasn’t ideal for the majority of human disease mutations, which result from errors known as point mutations.
Throughout the history of medicine, we have not known yet how to efficiently correct point mutations to change that disease-causing T back to a C, perhaps until now.
It can be done through base editing
CRISPR/Cas9 acts as scissors that cut both strands of DNA. As the cell repairs the break, random bases can be inserted or deleted, altering the gene sequence. Large segments might even be deleted or rearranged. As explained, this does not work in most types of cells and disrupted gene outcomes still predominate. Therefore a new technique called base editing is developed. It alters just a specific base using base editors, without making double-stranded cuts. Base editors cause fewer unwanted mistakes.
Chemical biologist David R. Liu researched methods to directly perform on an individual DNA base, to truly fix rather than disrupt the mutations that cause genetic diseases. The results of the efforts are molecular machines called base editors
Base editors use the searching mechanism of CRISPR. But after searching, instead of cutting the DNA, they directly convert one base to another base without disrupting the rest of the gene.
Base editors do not exist in nature. So how to make base editors?
It was started by taking CRISPR scissors and disabling the ability to cut DNA but retaining the CRISPR ability to search for and bind a target DNA sequence. To the CRISPR sequence, we attach a second protein that performs a chemical reaction on the DNA base C and converting it into a base that behaves like T. A third protein is attached to the first two proteins which protect the edited base from being removed by the cell.
The outcome is an engineered three-part protein that allows conversation of Cs into Ts at specified locations in the genome.
Can we cure genetic diseases?
All this advancement about base editors happened two and a half years ago, but even in that short time, base editing has become widely used by the biomedical research community.
Base editors have not entered human clinical trials but scientists have succeeded in achieving a critical milestone by using base editors in animals to correct point mutations that cause human genetic diseases.
In the agriculture industry, base editors have been used to introduce single DNA letter changes that could lead to better yielding crops. Biologists have used these to find the role of letters (A, T, G, C) in genes in connection with diseases such as cancer.
All these applications have taken place in less than three years. Base editing has given us the exciting new reality that has made scientists hopeful to make critical advances in patient care provided it is used ethically.
the article inspired by chemical biologist David R. Liu’s breakthrough.